An Integrated Platform for
Data Cleaning & Clinical Trial Design
Sigma BioAnalytics unifies data cleaning, sample size & trial design, and clinical trial intelligence into one coherent, secure workflow — built for regulated research.
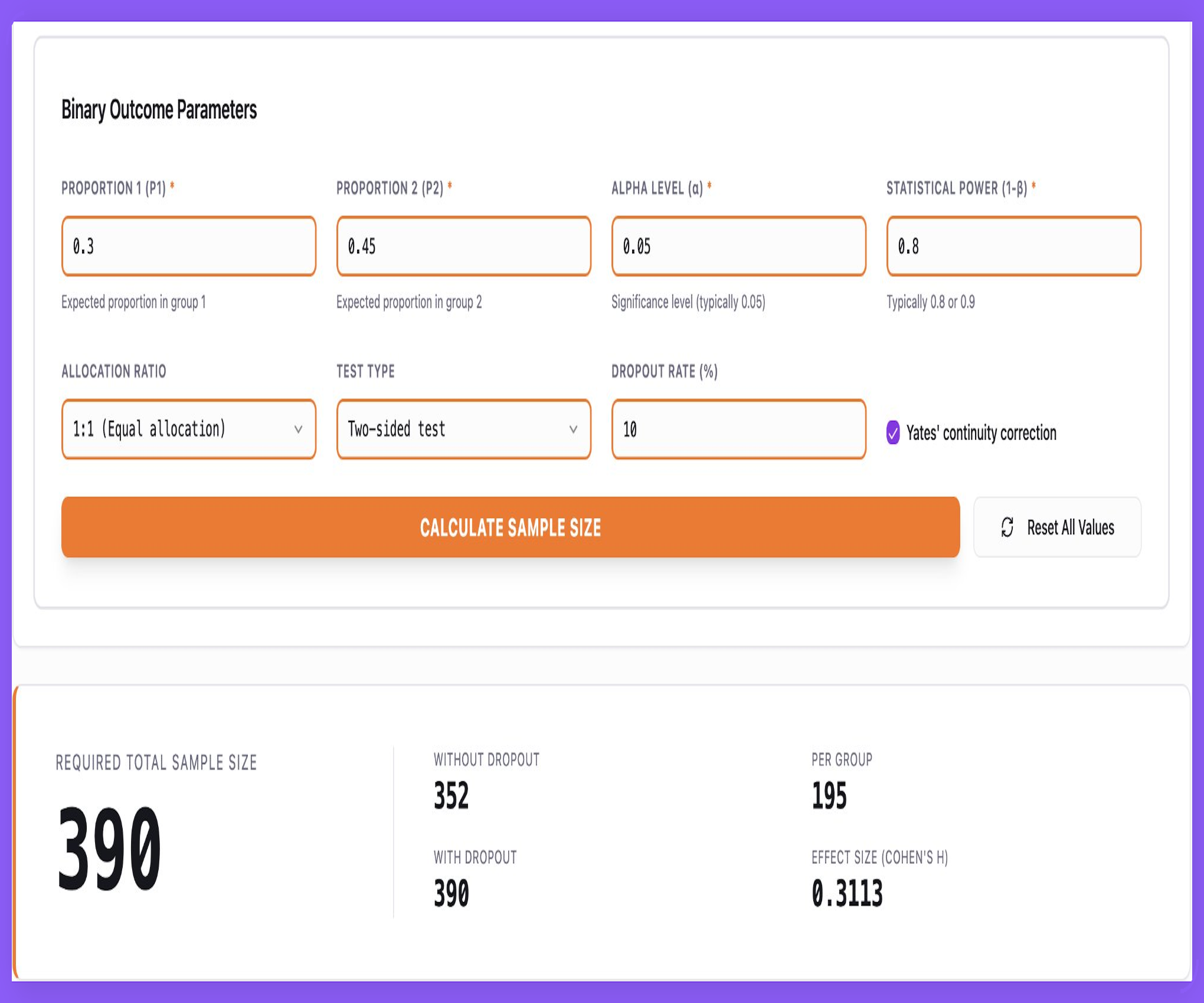
Validated Sample Size Calculations — Built In
Sigma provides production-grade statistical engines directly in the platform — eliminating spreadsheets, scripting, and undocumented assumptions, while remaining aligned with regulatory expectations.
- Binary, continuous, and time-to-event sample size calculations
- Superiority, non-inferiority, equivalence, and survival designs
- Dropout handling and sensitivity analyses included by default
- Protocol-ready outputs validated against PASS and nQuery
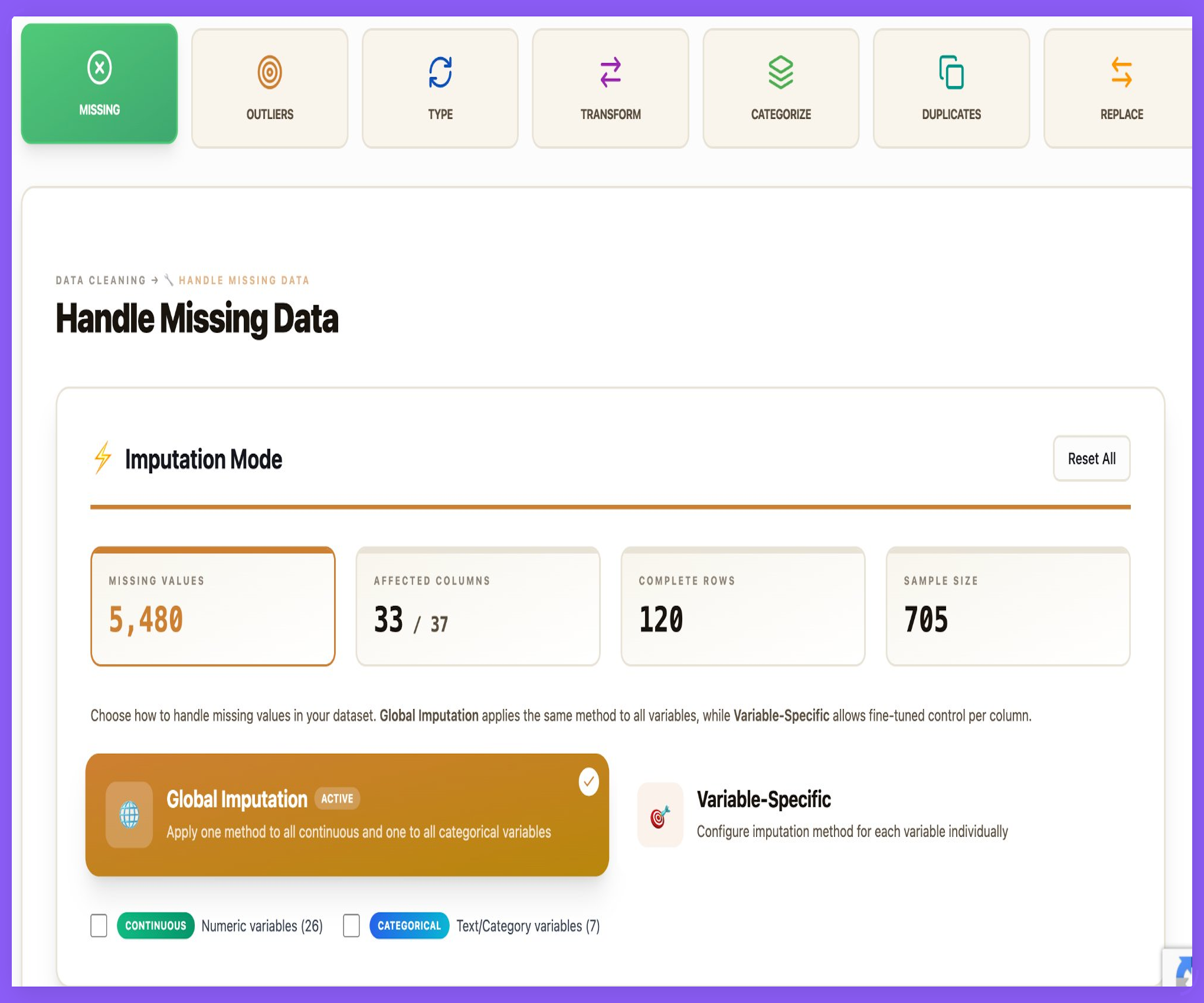
Transparent, Auditable Data Cleaning — Without Code
Sigma’s Data Cleaning Studio replaces spreadsheets and ad-hoc scripts with a guided, fully auditable workflow — designed for regulated clinical and observational research.
- Automated detection of missing data, outliers, and inconsistencies
- Global or variable-specific imputation strategies
- Clear visibility of affected columns and retained sample size
- Every transformation logged, traceable, and reproducible
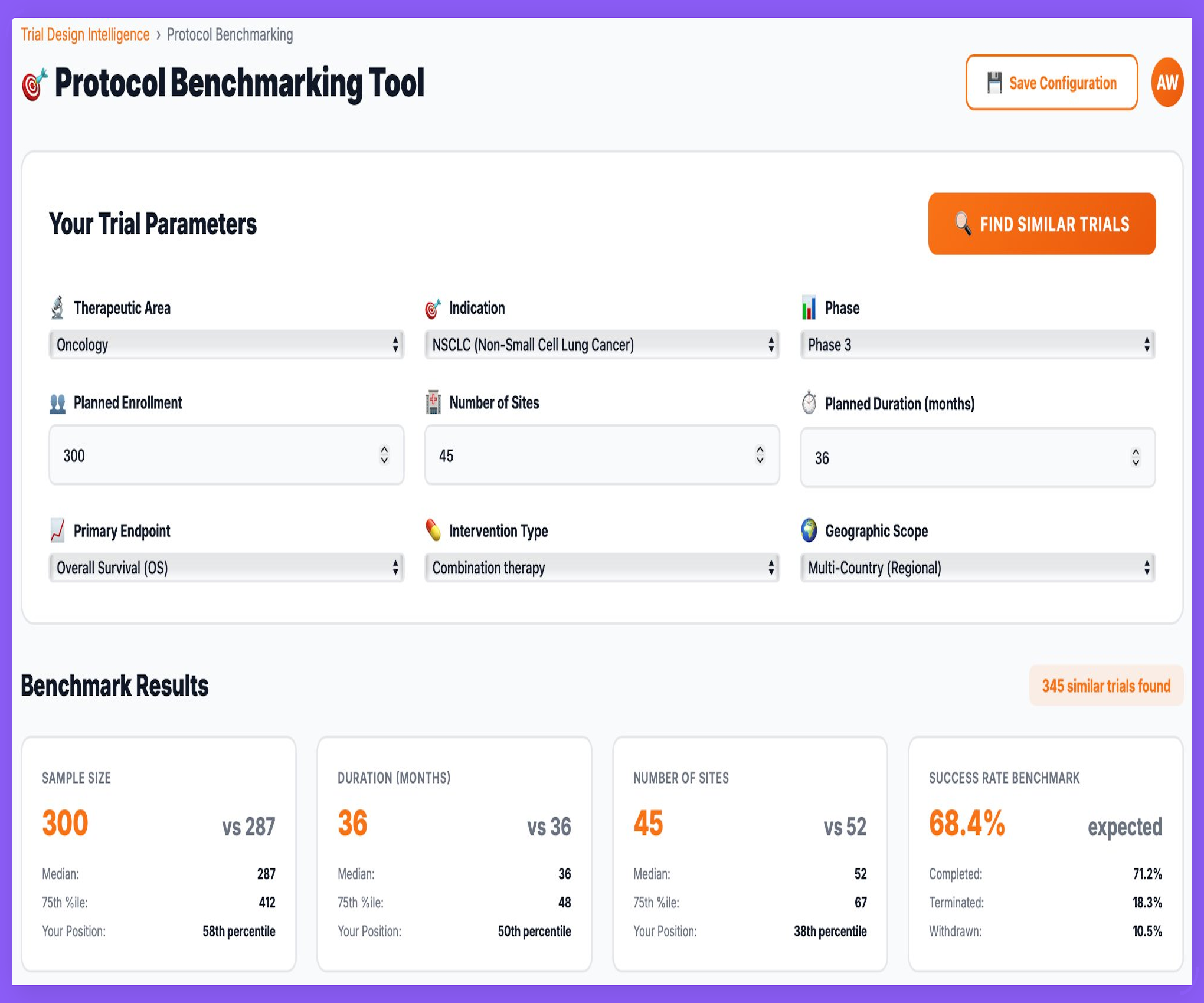
Protocol Benchmarking — Grounded in Real Trials
Sigma’s Clinical Trial Intelligence (CTI) allows teams to benchmark proposed trial designs against hundreds of historically similar studies — providing empirical context for feasibility, timelines, and risk.
- Benchmark enrolment, duration, and site count against comparable trials
- Position your design within historical percentiles and medians
- Review observed success, termination, and withdrawal rates
- Support protocol decisions with evidence — not assumptions
Built for Teams Working Where Decisions Matter
Sigma is designed for organisations that require rigor, traceability, and speed — without relying on fragmented tools or opaque workflows.
Biotech & Pharma Teams
CROs & Statistical Consultants
Academic & Translational Research
One Platform. Three Complementary Tools.
Sigma replaces fragmented workflows with a single, integrated environment designed around how clinical studies are actually planned and justified.
Integrated by Design
Data cleaning, study design, and clinical intelligence operate within one coherent analytical workflow.
Transparent & Auditable
Every step is visible, reproducible, and suitable for governance and regulatory discussion.
No-Code, Research-Grade
Guided workflows remove scripting overhead without sacrificing statistical rigor.
Three Tools. One Coherent Workflow.
Sigma’s tools are designed to complement each other — enabling teams to move seamlessly from raw data to trial-ready decisions.
Data Cleaning Studio
- Automated missingness, outlier, and duplication checks
- Transformations, categorisation, and replacements
- Full audit trail of all operations
- Export to standard statistical formats
Sample Size & Trial Design
- Binary, continuous, and time-to-event endpoints
- Superiority, non-inferiority, equivalence designs
- Sensitivity analyses for assumption testing
- Protocol-ready PDF and Word reports
Clinical Trial Intelligence
- Competitive landscape and benchmarking
- Endpoint and protocol intelligence
- Site and investigator feasibility insights
- Historical trial outcome analysis
Enterprise Capability — Without Enterprise Pricing
Sigma operates on a simple monthly subscription model, giving teams access to all tools at a fraction of the cost of traditional commercial software.
Simple Subscription
One monthly plan. No per-tool licenses. No long-term lock-ins. No hidden complexity.
Cost-Effective by Design
Access advanced statistical and intelligence tools without the overhead of legacy enterprise software.
Scales with Your Team
Suitable for early-stage biotechs, consultancies, academic groups, and growing clinical teams.
Secure. Ephemeral. Fully Controlled.
Sigma is built as a secure, multi-tenant analytics environment designed for confidential research and early-stage feasibility work.
Ephemeral by Default
No persistent data storage. Automatic secure deletion within 24 hours unless retained by the user.
Full User Ownership
Users retain complete ownership and responsibility for uploaded data and exported results.
No Internal Access
No internal system administrator access to client data. Sigma provides the environment — not data custody.
Ready to Modernise Your Clinical Analytics Workflow?
Whether you're planning early feasibility or supporting a full clinical programme, Sigma helps you move faster — with confidence and transparency.